
Revista Portuguesa de Estomatologia, Medicina Dentária e Cirurgia Maxilofacial
SPEMD - Revista Portuguesa de Estomatologia Medicina Dentária e Cirurgia Maxilofacial | 2025 | 66 (4) | 191-198
Original research
Effect of different cleansing agent solutions on denture acetal resin mechanical and physicochemical properties
Efeito de diferentes soluções de agentes de limpeza nas propriedades mecânicas e físico-químicas da resina acetal para próteses dentárias
a Department of Dental Technology, Medical University of Lodz, Lodz, Poland
b University Laboratory of Materials Research, Medical University of Lodz, Lodz, Poland
Zofia Magdalena Kula - zofia.kula@umed.lodz.pl
Article Info
Rev Port Estomatol Med Dent Cir Maxilofac
Volume - 66
Issue - 4
Original research
Pages - 191-198
Go to Volume
Article History
Received on 09/03/2025
Accepted on 12/12/2025
Available Online on 30/12/2025
Keywords
Original Research
Effect of different cleansing agent solutions on denture acetal resin mechanical and physicochemical properties
Efeito de diferentes soluções de agentes de limpeza nas propriedades mecânicas e físico-químicas da resina acetal para próteses dentárias
Zuzanna Reeves1 0009-0001-5903-688X
Agata Szczescio-Włodarczyk2 0000-0002-9633-5096
Zofia Magdalena Kula1 0000-0002-6899-9098
1 Department of Dental Technology, Medical University of Lodz, Lodz, Poland.
2 University Laboratory of Materials Research, Medical University of Lodz, Lodz, Poland
Article history:
Received 9 March 2025
Accepted 12 December 2025
Available online 31 December 2025
Abstract
Objectives: To determine the impact of denture hygiene solutions on the physicochemical properties of acetal material used for dental prosthesis production.
Methods: The study used T.S.M Acetal, immersed in denture hygiene solutions. The samples were divided into five groups: a control group, a group immersed in distilled water, and three groups immersed in a dental hygiene solution: Multiclean Roko powder for thermoplastics, Corega Tabs, and Protefix® Hygiene, respectively. After immersion, tests for hardness, impact strength, flexural strength, and sorption were conducted. Statistical analysis of the collected results was performed in Statistica 13.3. The normality of the distribution was checked using the Shapiro-Wilk test. When values were p>0.05, considered as a normal distribution, a parametric analysis of variance ANOVA followed by a post-hoc test was performed.
Results: After immersion of acetal in Corega Tabs and Protefix® Hygiene solutions, hardness significantly decreased. The unnotched impact strength test revealed an increase for all groups, whereas the notched impact strength decreased significantly, except for the Multiclean Roko solution group. Flexural strength after solution immersion did not differ significantly from the control group, except for the Corega Tabs group. The sorption study revealed a large decrease for the Corega Tabs (2,2 μg/mm3) and Protefix® Hygiene groups.
Conclusions: The Multiclean Roko cleaning agent for thermoplastic materials has the least impact on material hardness, so its use for the hygienization of acetyl-based restorations seems justified. The impact of other agents on thermoplastic material hardness might increase abrasive wear, which should be investigated in further studies.
Keywords:Acetal resin, Dental prosthesis, Dentures, Oral hygiene
Resumo
Objetivos: Determinar o impacto de soluções de higiene de próteses dentárias nas propriedades físico-químicas do material acetal utilizado no fabrico de próteses dentárias.
Métodos: O estudo utilizou o T.S.M Acetal, incubado em preparações para higiene de próteses dentárias. As amostras foram divididas em cinco grupos: um grupo controlo, um grupo incubado em água destilada e três grupos incubados em soluções para higiene de próteses dentárias: Multiclean Roko em pó dedicado a termoplásticos, Corega Tabs e Protefix® Hygiene, respetivamente. Após a incubação, foram realizados ensaios de dureza, resistência ao impacto, resistência à flexão e sorção.
Resultados: Após a incubação do acetal nas preparações Corega Tabs e Protefix® Hygiene, a dureza diminuiu significativamente. O teste de resistência ao impacto sem entalhe revelou um aumento em todos os grupos, enquanto a resistência ao impacto com entalhe mostrou uma diminuição significativa, exceto no grupo de Corega Tabs. A resistência à flexão após a imersão não diferiu significativamente da do grupo controlo, exceto no grupo Corega Tabs. O estudo de sorção revelou uma grande diminuição nos grupos de Corega Tabs e Protefix Hygiene.
Conclusões: O agente de limpeza para materiais termoplásticos Multiclean Roko tem o menor impacto na dureza do material, pelo que parece justificada a sua utilização na higienização de restaurações à base de acetilo. O impacto de outros agentes na dureza do material termoplástico poderá aumentar o desgaste abrasivo do material, o que deve ser investigado em estudos posteriores.
Palavras-chave: Resina acrílica, Prótese dentária, Próteses dentárias,Higiene oral
Introduction
The problem of complete or partial edentulism increases with na aging society.1 Tooth loss is commonly treated with removable dentures. An excellent alternative to the widely used methyl methacrylate and metal alloys in their fabrication is thermoplastic materials, from which flexible dentures are made. These restorations provide excellent aesthetics and user comfort.2
An example of a thermoplastic material is acetal. This polymer, also known as polyoxymethylene (POM), is obtained from formaldehyde. It is characterized by its flexibility, excelente aesthetics, shape stability, and impact strength. During the fabrication of dentures from this material, no chemical reactions occur, so the final work does not contain residual monomer.3, 4 However, acetal resin with acrylic teeth set during restoration production is connected to them only by mechanical bonding; no chemical bond is created between them, which may result in them breaking off.
Although acetal has many advantages, there are also some disadvantages to note. Despite their considerable color stability, products containing carotene may cause its discoloration.
Moreover, in cases of insufficient oral hygiene, clasps located deep in the undercuts may cause periodontal trauma and the development of periodontitis. On the other hand, the process of making a dental prosthesis using acetal material is fraught with additional difficulties for the dental technician. Namely, in the final stage of producing such a prosthesis, it isn’t easy to polish it and maintain the gloss, and special pastes should be used for this purpose. Moreover, it is important to remembre that this type of denture cannot be modified or repaired.
The surface structure of acetal resin is rougher than that of acrylic, which promotes faster accumulation of plaque and deposits, requiring the user to maintain meticulous hygiene.
For a prosthetic restoration to fulfill its proper function for a long time, its hygiene is essential. During denture use, similarly to the patient’s own teeth, discolorations occur, food residues accumulate, and a biofilm forms containing many microorganisms that can cause oral diseases and systemic illnesses.5, 7
For the hygiene of prosthetic restorations, both manual and chemical methods can be used. In the chemical method, the denture is immersed in a cleaning solution. This is an easy, quick, and cheap method, especially useful for patients with impaired motor coordination.7 However, these agents can affect the properties of the material that composes the restoration.
Therefore, it is very important to obtain information on the impact of hygiene preparations on thermoplastic materials.
The objective of this work was to determine the effect of denture hygiene solutions on selected properties (hardness, three-point bending strength, impact strength, and sorption) of acetal material used for making dental prostheses. The null hypothesis is that denture cleaning solutions do not affect the mechanical properties of thermoplastics.
Material and Methods
A total of 100 samples were made from T.S.M material (Pressing Dental, San Marino) by injection molding using an automatic injection molding machine J-100 (Pressing Dental, San Marino). The samples were divided into five groups:
• Group I – initial acetal sample,
• Group II – acetal immersed in distilled water,
• Group III – acetal immersed in Multiclean solution (Roko, Poland),
• Group IV – acetal immersed in Corega Tabs double-strength solution (Stafford-Miller, Ireland),
• Group V – acetal immersed in Protefix Hygiene solution (Queisser Pharma, Germany).
Each group contained 20 samples, divided into four subgroups for the four test types. The samples for hardness, impact strength, and sorption tests had dimensions of 20 mm x 10 mm x 5 mm, while the samples for bending strength tests had dimensions of 64 mm x 10 mm x 4 mm.
A simulation of 1 year of product use was performed, and the immersion time for each group was adjusted according to the manufacturer's recommendation, as follows:
• Group II – 7,300 minutes in distilled water,
• Group III – 7,300 minutes (20 minutes × 365 days) in the Multiclean Roko solution,
• Group IV – 1,460 minutes (4 minutes × 365 days) in Corega Tabs solution,
• Group V – 1,825 minutes (5 minutes × 365 days) in Protefix® Hygiene solution.
The solutions were prepared according to the manufacturer's instructions. The substance was dissolved in 250 ml of water and then poured over the samples placed in sealed glass containers. The samples were arranged to maximize the surface area in contact with the liquid. Incubation took place at room temperature. The solution of Multiclean Roko denture cleaning powder was replaced every 24 hours, and
the solution of Corega Tabs and Protefix Hygiene tablets every 4 hours.
After immersion, Vickers hardness testing, notched and unnotched impact strength testing using the Dynstat method, three-point bending strength testing, and sorption testing were conducted.
The hardness of the samples was tested using the Vickers method on three randomly selected samples intended for unnotched impact strength testing. A total of nine measurements were performed for each research group using a semi-automatic hardness tester (Roell ZHVμ, Zwick-Roell, Germany). The indenter load was set to 1 kg, and the loading time to 10 seconds.
The diagonals of the indentation were measured to calculate the Vickers hardness value.(Figure 6)
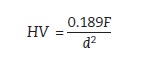
Figure 6:
F static load [N]
d – arithmetic mean of the two indentation diagonals d1 and d2 [mm]
Impact strength testing of 20 mm × 10 mm × 5 mm notched and unnotched samples was carried out using the Dynstat method with the Zwick/Roell HIT5.5 machine
Notched impact strength (an), unnotched impact strength (ak), and relative impact strength were calculated using the following formulas (figures 7, 8 and 9):
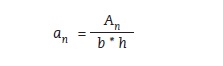
Figure 7:
An – work required to break the sample [J]
b – sample width [cm]
h – sample thickness [cm]
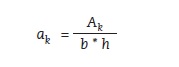
Figure 8:
Ak – work required to break the sample [J]
b – sample width [cm]
h – sample thickness [cm]
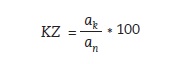
Figure 9:
KZ – relative impact strength
ak – unnotched impact strength
an – notched impact strength
Bending strength testing was conducted using the threepoint bending method with the Zwick/Roell Z020 strength testing machine (Zwick-Roell, Germany). The initial load was set to 0.5 MPa and the measuring head movement speed to 5 mm/min. Measurements were carried out on five samples per test group, all 25 samples of dimensions 64 mm × 10 mm × 4 mm. The support span was 40 mm. The three-point bending strength was calculated using the formula below (Figure 10):
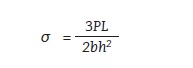
Figure 10:
• P – force acting on the sample [MPa]
• L – support span [mm]
• b – sample width [mm]
• h – sample thickness [mm]
For the sorption test, 20 samples of dimensions 20 mm × 10 mm × 5 mm were used—5 samples from groups II, III, IV, and V. Each sample was weighed on an analytical balance (RADWAG® model XA 82/220/X, Radwag, Poland) with a reading accuracy of 0.01/0.1 mg. After weighing, all samples were placed in a kiln, which was then placed in a vacuum laboratory dryer (model DZ-2BC II, ChemLand, Poland). After re-weighing and achieving a constant sample mass, the samples were immersed according to their group:
• Group II in distilled water,
• Group III in Multiclean Roko powder solution,
• Group IV in Corega Tabs tablet solution,
• Group V in Protefix® Hygiene solution.
After immersion, the samples were re-weighed and placed in a desiccator in a laboratory dryer at 37ºC. After achieving a constant sample mass after drying, the test was completed, and water sorption was calculated according to the formula below (Figure 11):
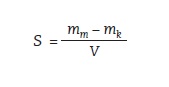
Figure 11:
• S– sorption
• mm – mass of wet sample
• mk – mass of dried sample
• V – sample volume
Statistical analysis of the results was conducted using the Statistica 13.3 program. The tested groups were independent.
In all cases, the normality of distribution was first checked using the Shapiro-Wilk test. A value of p>0.05 was considered indicative of normal distribution. If the distribution was normal, a parametric analysis of variance (ANOVA) was performed. Levene's test was used to check the homogeneity of variances; if p>0.05, ANOVA followed by a posthoc test was performed. In the case of a non-normal distribution, the Kruskal-Wallis test and a post-hoc test were conducted.
Results
Hardness testing (Figure 1) showed that the average hardness of the control sample was 18 on the Vickers scale; Group II samples kept in distilled water also achieved the same value. The lowest hardness value was obtained by Group IV, with samples kept in Corega Tabs solution: 14 on the Vickers scale.
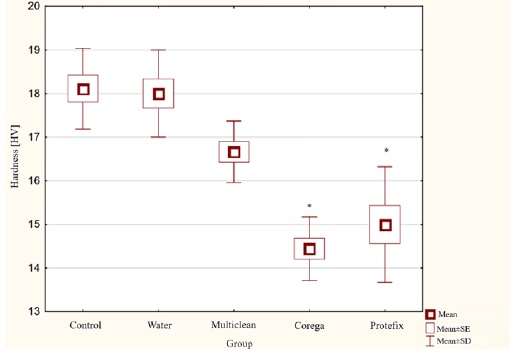
Figure 1. Results of the Vickers hardness test for acetal material subjected to aging in denture hygiene solutions.
For the hardness results, a statistical analysis was performed by first checking whether the distribution was consistent with a normal pattern.
The highest average value of unnotched impact strength (Figure 2) was recorded for Group IV acetal immersed in the Corega Tabs tablet solution: 4.63 J/cm². The lowest unnotched impact strength value was 3.13 J/cm², belonging to the control group. In the case of notched impact strength (Figure 3), the highest average impact strength was 1.76 J/cm² for Group III after immersion in the Multiclean Roko preparation solution, and the lowest was 0.88 J/cm² for Group IV after incubation in the Corega Tabs solution. Statistical analysis of the notched and unnotched impact strength results did not show significant differences between the tested groups (p>0.05).
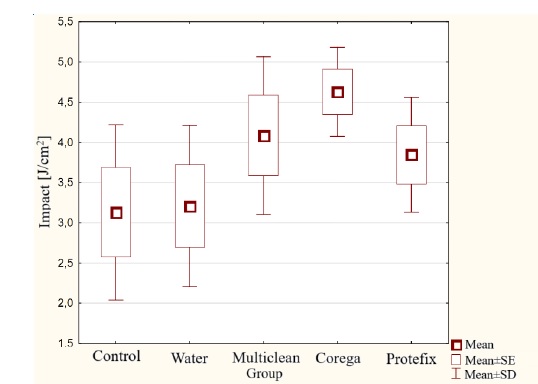
Figure 2. Unnotched impact strength results for acetal material subjected to aging in denture hygiene solutions.
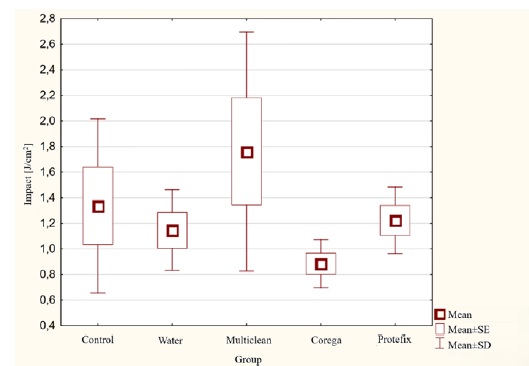
Figure 3. Notched impact strength results for acetal material subjected to aging in denture hygiene solutions.
The three-point bending strength measurement (Figure 4) showed the highest value for Group IV immersed in the Corega Tabs solution, at 111.6 MPa. The lowest result was recorded for the control group: 102.1 MPa. However, no statistically significant differences were found between the tested groups.
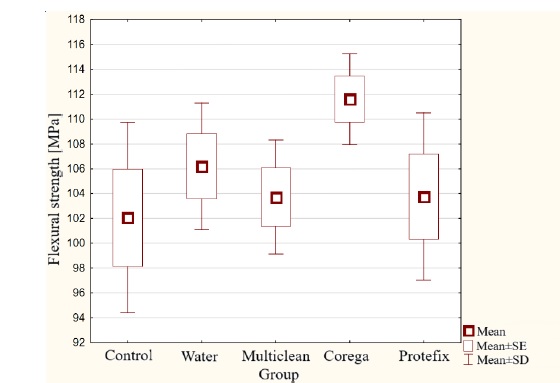
Figure 4. Bending strength results for acetal material subjected to aging in denture hygiene solutions.
The sorption test results showed the highest value, 4.2 μg/mm³, for samples from Group II immersed in water. The lowest result, 2.2 μg/mm³, was obtained by Group IV samples immersed in the Corega Tabs solution. Statistically significant differences were found between groups stored in Corega and Protefix solutions and groups of samples immersed in water and Multiclean solution. Clear differences between these groups are shown in Figure 5.
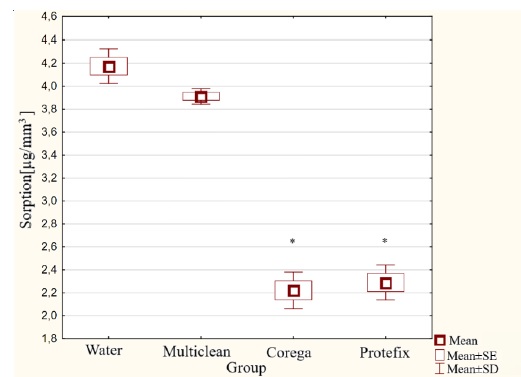
Figure 5. Sorption results for acetal material aged in solutions of denture hygienization preparations.
Discussion
There are many agents available on the market that significantly facilitate the removal of denture contaminants formed during their use. It should be noted, however, that these are chemical substances that can affect the material from which dentures are made.
In 2013, Polyzois et al.8 conducted studies on the effect of denture hygiene solutions on the sorption of acetal material.
In their work, they clearly emphasized the need to also examine the material's mechanical properties, such as bending strength, hardness, and impact strength.
In the present study, the results of hardness testing on the Vickers scale indicated lower hardness values for materials immersed in Corega Tabs (14 HV) and Protefix® Hygiene (15 HV) solutions compared to the control and samples immersed in water. Molka et al.9 obtained similar hardness results for samples immersed in Protefix solution: approximately 17 HV.
In turn, the Multiclean Roko solution, intended for the hygiene of dentures made of thermoplastic material, showed a small decrease in hardness.10 Faiza et al.11 reported that componentes of denture hygiene agents penetrate between polymer chains, contributing to a decrease in material hardness.
Porojan et al.10 pointed out that water absorption by polymers contributes to hydrolytic degradation, leading to changes in the thermoplastic material's structure and properties. Deterioration of the hardness of the material from which the denture is made can cause abrasion of the material's surfasse during brushing. The resulting scratches will promote the deposition of biofilm and food residues, resulting in poor aesthetics of the restoration.
A decrease in the material's ability to withstand sudden loads means that the denture will be more susceptible to damage during daily use, such as by accidental drops. The impact strength test revealed increases in unnotched impact strength for all groups immersed in denture hygiene solutions (Groups III, IV, V). On the other hand, notched impact strength increased compared to the control group only for the samples stored in Multiclean Roko solution. The relative impact strength was calculated for each test group. In the control group, it was 42.7%, while for the group of samples immersed in water it was 35.8%. The highest value was obtained for the group immersed in the Multiclean Roko powder solution, 43.1%, and the lowest, 19.1%, for the group immersed in the Corega Tabs solution. This indicates that the impact strength of acetal decreases significantly when simulating material defects in the form of a notch. Weakening after dynamic impacts may be caused by the polymer network absorbing inorganic and organic compounds from the solutions of denture hygiene preparations, as mentioned by Faiza et al.11 These researchers also explain that water penetration between polymer chains leads to swelling, thereby decreasing the material's mechanical properties.12 - 14
In the bending strength test, all groups showed higher values than the control sample. The greatest difference occurred in the group immersed in the Corega Tabs solution: 111.6 MPa. Groups immersed in Multiclean Roko and Protefix® Hygiene solutions had nearly identical values: 103.7 MPa and 103.8 MPa, respectively.
Sorption of materials placed in Corega Tabs and Protefix® Hygiene solutions was significantly lower than that of materials immersed in water and Multiclean Roko solution. Lower water sorption is favorable for the material from which the denture is made, as it is associated with reduced fluid absorption from the oral environment, reducing the likelihood of dimensional changes or weakening of the material's properties.
Researchers also point out decreased biofilm deposition, especially the adhesion of Candida albicans.15 During the sorption test, the group immersed in distilled water showed na average sorption value of 4.2 μg/mm³. In the study by Molka et al.,9 this value was approximately 8.5 μg/mm³, and Polyzois et al.8 obtained a result of 10.65 μg/mm³.10 The more than twofold difference can be explained by the different sample immersion times adopted: 7,300 minutes in this study, 5,475 minutes in Molka et al.,9 and 100 days of sample immersion in water at room temperature in Polyzois et al.8. In this study, samples immersed in Protefix® Hygiene tablets obtained a result of 2.3 μg/mm³, which again differs from the result by Molka et al.9: 6.03 μg/mm³. Polyzois et al.8 also studied sorption after storing samples in Corega solution, obtaining a result of 9.39 μg/mm³, which is much higher than the result of 2.2 μg/mm³ obtained in the present study. The results cannot be directly compared due to different incubation times, methodologies, and dimensions: Polyzois et al.8 placed the samples in the solution for 8 hours per day and then stored them in water, and used samples measuring 38 mm × 38 mm × 1.8 mm; in this study, the samples had dimensions of 20 mm × 10 mm × 5 mm. Despite these differences, the results of this study and those by Molka et al.9 and Polyzois et al.8 do not exceed the maximum permissible sorption value indicated by the standard PN-EN ISO 1567:2002, which is 32 μg/mm³.
The present study had several limitations, including limited material availability. The limited number of samples increased the risk of random errors affecting the statistics. Another major limitation was the duration of the study: 1 year of denture hygiene agent use was simulated, with solutions replaced every 12 hours and no breaks between immersions. Our simulation may contain errors due to the significant difficulty of conducting an identical simulation that accurately reflects the prosthesis cleaning process. This is difficult because the denture is placed in the solution only for a short period, usually a few minutes, and then removed. Finally, the difference between individual rounds of sample-taking and the continuous maintenance required by the injection molding machine also limited the overall confidence level.
Conclusions
The used agents do not affect the strength properties of the acetal material, such as impact strength and three-point bending strength. However, immersion in certain denture hygiene agents affects the material's hardness, potentially increasing abrasive wear. The cleaning agent for thermoplastic materials has the least impact on material hardness. Finally, sorption of materials placed in Corega Tabs and Protefix® Hygiene solutions was significantly lower than that of materials after immersion in water and Multiclean Roko solutions.
References
1. Amjad CM, Azad AA, Ayub MM, Qureshi MA, Javed MU. Denture hygiene habits in complete denture wearers at armed forces institute of dentistry. Pak Armed Forces Med J. 2010;60(4). Available from: https://pafmj.org/PAFMJ/article/view/1521. Accessed: 9 Mar, 2025.
2. Mekkawy MA, Hussein LA, Alsharawy MA. Comparative study of surface roughness between polyamide, thermoplastic polymethyl methacrylate and acetal resins flexible denture base materials before and after polishing. Life Sci J. 2015;12:90-5.
3. Alagwany A, Fatoh D, Abbas M, Helal MA, Mahmoud II. Wear Resistance Evaluation of the Thermoplastic. Journal of Clinical Research in Dentistry. 2019; 2(2):1-5.
4. Kowalski R, Kozak BM, Sobolewska E. Contemporary hybrid acrylic materials and modern thermoplastics in the manufacture of dental prostheses. Pomeranian J Life Sci. 2023;69:39-45.
5. Oussama M, Ahmad H. Materials and methods for cleaning dentures a review. International Journal of Dental Clinics. 2014;6(2):19-22.
6. Pawela J, Szczesio-Włodarczyk A, Kula ZM. Effect of denture cleansers on the properties of acrylic base material. Protet Stomatol. 2020;70:375-81.
7. Barochia J, Kamath S. Evaluation of the effect of denture cleansers on the surface roughness of hard denture base material: An in vitro study. Indian J Dent Res. 2018;29:657-62.
8. Polyzois G, Niarchou A, Ntala P, Pantopoulos A, Frangou M. The effect of immersion cleansers on gloss, colour and sorption of acetal denture base material. Gerodontology. 2013;30:150-6.
9. Molka A, Szczesio-Włodarczyk A, Dworak M, Kula ZM. Influence of the solution of tablets for denture hygiene on the mechanical and physicochemical properties of various polymeric materials used in the manufacture of dental prostheses. Protet Stomatol. 2024;74:16-24.
10. Porojan L, Vasiliu RD, Porojan SD, Bîrdeanu MI. Surface quality evaluation of removable thermoplastic dental appliances related to staining beverages and cleaning agents. Polymers (Basel). 2020;12:1736.
11. Faiza A, Fauzia Q, Akram S. Impact strength of acrylic resins after storage in denture cleansers. Pakistan Oral & Dental Journal. 2014;34:735-8.
12. Nascimento I, dos Santos NR, Anes V, Neves CB, Santos V. Effect of Thermal Aging and Chemical Disinfection on the Microhardness and Flexural Strength of Flexible Resins. Appl Sci. 2024;14:361.
13. Sampaio-Fernandes M, Ferreira Júnior C, Oliveira SJ, Martins RC, Sampaio-Fernandes JC, Figueiral MH. In Vitro Comparative Study of the Surface Properties of Materials for Removable Prosthetic Bases. Rev Port Estomatol Med Dent Cir Maxilofac. 2022;63:117-25.
14. Costa J, Matos A, Bettencourt A, Portugal J, Neves CB. Effect of Ethanol Solutions as Post-Polymerization Treatment on the Properties of Acrylic Reline Resins. Rev Port Estomatol Med Dent Cir Maxilofac. 2016;57:215-22.
15. Fathy SM, Abdel-Halim MS, El-Safty S, El-Ganiny AM. Evaluation of polymethyl-methacrylate and acetal denture base resins processed by two different techniques before and after nano-chlorohexidine surface treatment. BMC Oral Health. 2023;23:985.
Zofia Magdalena Kula
E-mail address: zofia.kula@umed.lodz.pl
CRediT authorship contribution statement
Zuzanna Reeves: Data curation, Formal analysis Investigation, Resources, Software, Validation, Visualization, Writing – original draft. Agata Szczescio-Włodarczyk: Data curation, Formal analysis, Methodology. Zofia Magdalena Kula: Conceptualization, Methodology, Project administration, Supervision, Validation, Visualization, Writing – review & editing.
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical disclosures
Protection of human and animal subjects. The authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of data. The authors declare that no patient data appear in this article.
Right to privacy and informed consent. The authors declare that no patient data appear in this article.
1646-2890/© 2025 Sociedade Portuguesa de Estomatologia e Medicina Dentária. Published by SPEMD.
This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
