
Revista Portuguesa de Estomatologia, Medicina Dentária e Cirurgia Maxilofacial
SPEMD - Rev Port Estomatol Med Dent Cir Maxilofac | 2018 | 59 (2) | 115-118
Case report
Head and neck giant keratoacanthoma: a case rep
Queratoacantoma gigante da cabeça e pescoço: caso clínico
a Oral Surgery and Pathology Department, João de Barros Barreto University Hospital, Federal University of Pará, Belém, Pará, Brazil
b Division of Oral and Maxillofacial Surgery, Dental School at Araraquara, UNESP, Araraquara, Brazil
Lucas Lacerda de Souza - lucaslac@hotmail.com
Article Info
Rev Port Estomatol Med Dent Cir Maxilofac
Volume - 59
Issue - 2
Case report
Pages - 115-118
Go to Volume
Article History
Received on 23/12/2017
Accepted on 04/08/2018
Available Online on 10/09/2018
Keywords
Case report
Head and neck giant keratoacanthoma: a case report
Queratoacantoma gigante da cabeça e pescoço: caso clínico
Yuri Edward de Souza Damascenoa, Waldner Ricardo Souza de Carvalhoa, Nicolau Conte Netoa, Eduardo Hochuli Vieirab, Lucas Lacerda de Souzaa,*, Hélder Antônio Rebelo Pontesa
a Oral Surgery and Pathology Department, João de Barros Barreto University Hospital, Federal University of Pará, Belém, Pará, Brazil.
b Division of Oral and Maxillofacial Surgery, Dental School at Araraquara, UNESP, Araraquara, Brazil.
http://doi.org/10.24873/j.rpemd.2018.09.227
Abstract
Keratoacanthoma is a low-grade tumor originated from the pilosebaceous glands that nearly resembles squamous cell carcinoma under histopathological analysis. Giant keratoacanthoma is a rare subtype with a size exceeding 2 cm, with a high aggressiveness potential. Head and neck giant keratoacanthomas are uncommon and mainly affect male patients with a mean age of 59.1 years old, usually affecting the nose. Excision of the lesion is the preferred treatment for giant keratoacanthoma. The aim of this study is to analyze the clinicopathological characteristics of head and neck giant keratoacanthoma by reviewing the previously published English literature and reporting one new case of a rare lesion affecting the lower lip.
Keywords: Excision, Giant keratoacanthoma, Treatment
Resumo
O queratoacantoma é um tumor de baixo grau, originário das glândulas pilossebáceas e que apresenta características do carcinoma de células escamosas sob análise histopatológica. O queratoacantoma gigante é um subtipo raro com um tamanho superior a 2 cm, com alto potencial de agressividade. O queratoacantoma gigante de cabeça e pescoço é incomum e afeta, principalmente, pacientes com idade média de 59,1 anos, com predileção para pacientes do sexo masculino e geralmente afeta o nariz. A excisão da lesão é o tratamento preferido para queratoacantoma gigante. O objetivo deste estudo é analisar as características clinicopatológicas do queratoacantoma gigante de cabeça e pescoço, revisando os estudos publicados na literatura inglesa e relatando um novo caso da lesão que apareceu no lábio.
Palavras-chave: Excisão, Queratoacantoma gigante, Tratamento
Introduction
Keratoacanthoma (KA) is a rapid growth tumor characterized by its composition of well-differentiated keratinizing squamous cells, which originate from the pilosebaceous follicles, and its etiology is not identified. 1, 4 It is commonly presented as a self-limiting lesion and has strong clinical and histopathological similarities with squamous cell carcinoma. 2, 3, 6
Head and neck KAs are common and account for around 70% of all KAs. 1, 3 Classically, the lesion is presented as a domeshaped nodule with an ulcerated crater in its center.6 Giant KA is a rare variant of KA with a size exceeding 2 cm. 1, 7 It has a main predilection for the nose and eyelid as primary sites8 occurs more commonly in men, and most of the lesions are observed in sun-exposed skin. 1, 4, 6
Owing to the rarity of the head and neck giant KA, few case reports and clinical series have been published describing its biological behavior on the head and neck region.
The aim of this work is to analyze the clinicopathological characteristics of the head and neck keratoacanthoma by reviewing all the case reports and clinical series previously published in English and reporting a rare new case in the lower lip.
Case report
A 64-year-old male patient was referred to the oral pathology service of the João de Barros Barreto University Hospital, Belém, Brazil, chiefly complaining of a 1-month rapidly growing swelling on his lower lip. The patient had a 1-year history of a lesion in the lower lip when, in the previous 4 weeks, he noticed a rapidly growing swelling and sought specialized assistance.
The patient had no history of trauma, infection, tumors or prior radiation. His medical history was noncontributory.
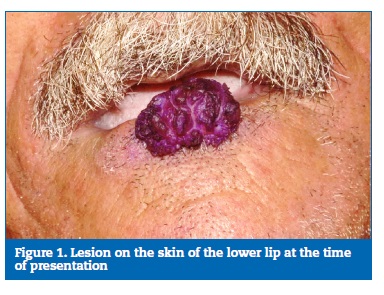
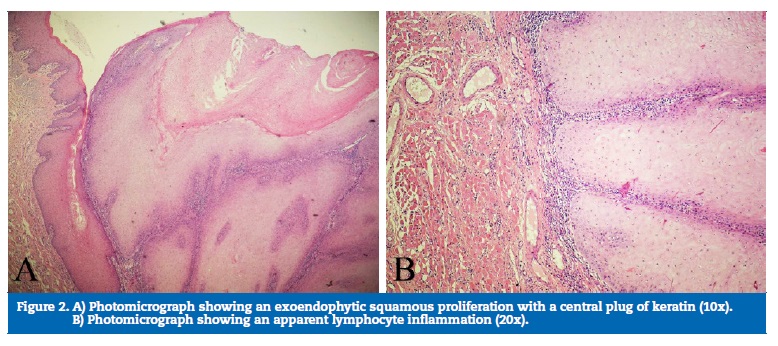
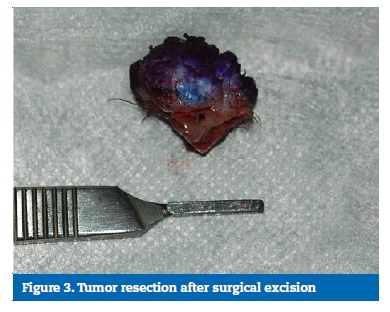
Physical examination showed no abnormalities. No lymph node infarction was observed in the head and neck after palpation. Clinically, the lesion exhibited an exophytic and pedunculated nodular appearance, with a verrucous surface and a violet coloration, measuring 3.2 x 2.5 in diameter (Figure 1). No lymph nodes were observed. The patient did not refer pain in the area, and the surrounding mucosa had a regular appearance. An incisional biopsy was performed under local anesthesia, and the histopathological exam revealed tumor islands with enlarged keratinocytes formed by a downgrowth of squamous epithelium with an increased amount of central keratin. The epidermis protruded irregularly and deeply into the dermis. Lymphocyte infiltration in the dermis was also observed (Figure 2). The suggestive diagnosis of KA was made due to the histopathological features. Then, the lesion was excised with a 1-mm margin, and the histopathological exam of the whole lesion led to the diagnosis of KA (Figure 3). No additional surgery was performed to establish a surgical margin because the diagnostic hypothesis was of KA, which led us to make a margin during the surgical procedures. The patient was lost for follow-up.
Discussion
KA was first described by Hutchinson in 1889.8 However, very few case reports and clinical series concerning the clinicopathological features of the head and neck giant KA have been published in the literature. In this study, the authors reviewed all case reports and clinical series dealing with the giant KA of the head and neck region, summarizing the most important aspects found by each author. A search in the Pubmed database revealed 47 cases since 1958, when the first case report on the head and neck giant KA was first published, presenting a 57-year-old male patient with a lesion in the eyebrow region.9
KAs in the head and neck region presenting more than 2 cm in diameter are rare and denominated giant KA. 1, 10 ‑ 11 In our study, we observed that the head and neck giant KA currently presents a mean size of 3.93 cm in diameter. Patients are usually in the sixth and seventh decade of life, with a mean age of 59.1 years old, and are mainly males. 1, 4, 6, 12, 13 Studies have reported that those tumors have a predilection for cosmetically sensitive areas, including the nose and the eyelids. 8, 14 Based on our literature analysis, the nose, the cheek and the lower lip are the locations most commonly affected by the head and neck giant KA.
The histopathological diagnosis of KA is further complicated by its histological similarities with the squamous cell carcinoma. 2, 3, 5 Histopathological features vary with the stage of evolution.3 An early lesion is reported to increase rapidly, generating cell differentiation with a high proliferation index.
The analysis varies according to the atypical mitosis, loss of polarity, individual cell keratinization and hyperchromatic cells of the specimen. 2, 3, 11 It is possibly associated with the time of evolution. The KA is a lesion characterized by a rapid and progressive growth. 4, 6, 11, 15 Our analysis revealed a mean evolution time of 15.1 months, although 73.3% of the cases presented a rapid progression in less than 3 days. Thus, while tumor progression occurs, the degree of cell proliferation increases and, consequently, it becomes increasingly similar to the squamous cell carcinoma. 2, 7 In the case report here presented, the surgical specimen was reviewed carefully, confirming the diagnosis of giant KA.
Numerous treatment methods have been used in patients with giant KA. Due to the tendency of KA to spontaneously regress, the efficiency of some treatments is difficult to evaluate. 5, 7, 12, 14, 16 Only 12.5% of the patients were followed solely by observation, and all of them presented total tumor regression after long follow‑up times. When no type of treatment is employed, there is a risk of the lesion invading adjacent tissues and aesthetic commitment. 2, 7 The non‑surgical treatments mainly used for the regression of the lesion were the intralesional interferon alpha 2 a17 and methotrexate. 6, 18 Taking into account the aggressive biological behavior of the head and neck giant KA, surgical excision was the most used treatment approach (29.1%), followed by combined surgery and radiation (8.3%). All patients showed tumor regression with no signs of recurrence, except in a case report where the treatment employed consisted of electrodesiccation and curettage associated with radiotherapy and bleomycin.19 The notable aggressive behavior of the lesion is confirmed by the occurrence of metastasis. A case of a 61‑year‑old man with lymph nodes, lungs and mediastinal metastasis has previously been reported.2 This aggravation may be associated with the lesion evolution time since the patient sought specialized treatment after 325 months of progression.
The degree of cell proliferation increases during tumor progression and, consequently, there is a greater invasive characteristic, similar to the squamous cell carcinoma. 2, 7 According to our literature review, the mean follow‑ up for the head and neck giant KA is 56.65 months, ranging from 2 to 240 months). This range may be explained by the treatment used since patients who underwent surgical procedures had a shorter treatment time and a fast resolution, 3, 13, 15, 20 - 22 while patients treated with intralesional therapy required multiple injections of the substances, resulting in longer follow‑up times. 6, 11, 17, 23 Typically, KA will recede spontaneously but may recur years later.31,32
In conclusion, according to our literature review, the head and neck giant KA is usually a rapid growth benign tumor with a low propensity for recurrence or metastasis that presents a predilection for male patients, affects mostly the nose and is commonly treated by surgery alone. The case report here presented revealed that tumor excision is an effective treatment and aesthetic principles need to be taken into account to have an optimal treatment and patient satisfaction.
References
1. Bogner PN, Cheney RT, Zeitouni NC. Giant keratoacanthoma: case report and review of the English literature. The American Journal of Dermatopathology. 2014;36:252‑7.
2. Alyahya GA, Heegaard S, Prause JU. Malignant changes in a giant orbital keratoacanthoma developing over 25 years. Acta Ophthalmol Scand. 2000;78:223-5.
3. Moriyama M, Watanabe T, Sakamoto N, et al. A case of giant keratoacanthoma of the auricle. Auris Nasus Larynx. 2000;27:185-8.
4. Yang Y, Xu Y, Wang L, Liao W, Gao T, Ma C. Two cases of giant keratoacanthoma centrifugum marginatum accompanied by α‐human papillomavirus infection. International journal of dermatology. 2015;54:951‑4.
5. de Visscher JG, van der Wal JE, Starink TM, et al. Giant keratoacanthoma of the lower lip. Report of a case of spontaneous regression. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1996;81:193-6.
6. de Visscher JG, van der Wal KG, Blanken R, Willemse F. Treatment of giant keratoacanthoma of the skin of the lower lip with intralesional methotrexate: a case report. J Oral Maxillofac Surg. 2002;60:93-5.
7. Shah M, Green L, Kwok C. Self‐resolving invasive squamous cell carcinoma or giant keratoacanthoma?. Clinical and experimental dermatology. 2012;37:701‑2.
8. Leonard AL, Hanke CW. Treatment of giant keratoacanthoma with intralesional 5‑fluorouracil. J Drugs Dermatol. 2006;5:454-6.
9. Kallos A. Giant keratoacanthoma. AMA Arch Derm. 1958;78:207-9. 10. Saftic M, Batinac T, Zamolo G, et al. HPV 6‑positive giant keratoacanthoma in an immunocompetent patient. Tumori. 2006;92:79-82.
10. Anadolu R, Akay BN, Bodamyali P, et al. Giant keratoacanthoma‑type low‑grade squamous cell carcinoma of the upper lip: response to intralesional interferon alpha‑2B. J Dermatolog Treat. 2011;22:239-40.
11. Lucente FE. Giant keratoacanthoma of the nose. Otolaryngol Head Neck Surg. 1985;93:112-6.
12. Phiske MM, Avhad G, Jerajani, HR. Keratoacanthoma centrifugum marginatum at an unusual site. Indian journal of dermatology. 2013;58:74.
13. Phiske MM, Avhad G, Jerajani, HR. Keratoacanthoma centrifugum marginatum at an unusual site. Indian journal of dermatology. 2013;58:74.
14. Browne F, O’Connell M, Merchant W, et al. Spontaneous resolution of a giant keratoacanthoma penetrating through the nose. Clin Exp Dermatol. 2011;36:369-71.
15. Edelman BA, Jacobs JB, Rotterdam H, et al. Giant keratoacanthoma: an atypical presentation. Otolaryngol Head Neck Surg. 1990;103:472-5.
16. Saito M, Sasaki Y, Yamazaki N, et al. Self‑involution of giant keratoacanthoma on the tip of the nose. Plast Reconstr Surg. 2003;111:1561-2.
17. Grob JJ, Suzini F, Richard MA, et al. Large keratoacanthomas treated with intralesional interferon alfa‑2a. J Am Acad Dermatol. 1993;29:237-41.
18. Spieth K, Gille J, Kaufmann R. Intralesional methotrexate as effective treatment in solitary giant keratoacanthoma of the lower lip. Dermatology. 2000;200:317-9.
19. Rapaport J. Giant keratoacanthoma of the nose. Arch Dermatol. 1975;111:73-5.
20. Pagani WA, Lorenzi G, Lorusso D. Surgical treatment for aggressive giant keratoacanthoma of the face. J Dermatol Surg Oncol. 1986;12:282-4.
21. Salam MA, Bickerton RC. Giant keratoacanthoma of the nasal vestibule. Br J Clin Pract. 1995;49:52.
22. Guimarães EP, de Carli ML, Sperandio FF, Pereira AA, Hanemann JA. Giant keratoacanthoma of the lower lip: case report and review of the published literature. General Dent. 2016;64:22‑5.
23. Santoso‑Pham JC, Shelley ED, Shelley WB. Aggressive giant keratoacanthoma of the face treated with intramuscular methotrexate and triamcinolone acetonide. Cutis. 1997;59:329-32.
24. Kyrmizakis DE, Hajiioannou JK, Helidonis E. Giant keratoacanthoma of the lower lip. Otolaryngol Head Neck Surg. 2003;128:904-5.
25. Hofer SO, Jackson IT. Self‑involution of giant keratoacanthoma on the tip of the nose. Plastic and reconstructive surgery. 2004;113:765‑6.
26. Tsikeloudi M, Lallas A, Sidiropoulos T, Chasioti V, Lefaki I, Apalla Z. Imiquimod as neoadjuvant treatment for giant keratoacanthoma arising on a background of lupus vulgaris. Clinical and experimental dermatology. 2014;39:60‑2.
27. Bart RS, Popkin GL, Kopf AW, et al. Giant keratoacanthoma: a problem in diagnosis and management. J Dermatol Surg. 1975;1:49-55.
28. Goldschmidt H, Sherwin WK. Radiation therapy of giant aggressive keratoacanthomas. Arch Dermatol. 1993;129:1162-5.
29. Kopf AW, Bart RS. Tumor conference no. 17: giant keratoacanthoma. J Dermatol Surg Oncol. 1978;4:444-5.
30. Liebman EP, Hauslein P. Giant keratoacanthoma of the ear. Trans Pa Acad Ophthalmol Otolaryngol. 1977;30:197-202.
31. Karaa A, Khachemoune A. Keratoacanthoma: a tumor in search of a classification. Int J Dermatol. 2007;46:671‑8.
32. Savage JA, Maize JC Sr. Keratoacanthoma clinical behavior: asystematic review. Am J Dermatopathol. 2014;36:422‑9.
Lucas Lacerda de Souza
E-mail address: lucaslac@hotmail.com
Ethical disclosures
Protection of human and animal subjects. The authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of data. The authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consent. The authors have obtained the written informed consent of the patients or subjects mentioned in the article. The corresponding author is in possession of this document.
Conflicts of interest
The authors have no conflicts of interest to declare.
Article history:
Received 23 December 2017
Accepted 4 August 2018
Available online 10 September 2018
